When clinical and laboratory findings suggest the diagnosis of MPS III, molecular genetic testing approaches can include use of a multigene panel.Īn MPS III or mucopolysaccharidosis multigene panel that includes GNS, HGSNAT, NAGLU, SGSH, and other genes of interest (see Differential Diagnosis) is most likely to identify the genetic cause of the condition at the most reasonable cost while limiting identification of variants of uncertain significance and pathogenic variants in genes that do not explain the underlying phenotype. Therapies under investigation: Despite ongoing research for a variety of therapeutic options, no treatments are currently clinically available for treatment of the primary manifestations of MPS III. Surveillance: Routine monitoring of: developmental capabilities and educational needs, destructive or disruptive behaviors musculoskeletal involvement hearing cardiac involvement.Īgents/circumstances to avoid: Procedures requiring anesthesia in centers ill-equipped or inexperienced in caring for patients with complex airway-management issues hip surgery (due to high risk of osteonecrosis of the femoral head) environments not adapted to minimize risk from unpredictable behaviors. Treatment of manifestations: Supportive therapies for neurodevelopmental delays, hearing loss, and visual impairment medications (rather than behavioral therapy) for psychiatric/behavioral issues physical therapy and/or orthopedic management of musculoskeletal manifestations and management as prescribed by consulting specialists for seizures, cardiac involvement, sleep disorders, feeding difficulties. Death usually occurs in the second or third decade of life secondary to neurologic regression or respiratory tract infections. Neurologic decline is seen in all affected individuals however, clinical severity varies within and among the four MPS III subtypes (defined by the enzyme involved) and even among members of the same family. Systemic manifestations can include musculoskeletal problems (joint stiffness, contractures, scoliosis, and hip dysplasia), hearing loss, respiratory tract and sinopulmonary infections, and cardiac disease (valvular thickening, defects in the cardiac conduction system). Disease course may be rapidly or slowly progressive some individuals with an extremely attenuated disease course present in mid-to-late adulthood with early-onset dementia with or without a history of ID. Disease onset is typically before age ten years. These investigative treatments are critical to address the limitations in treatment of the central nervous system (CNS).Mucopolysaccharidosis type III (MPS III) is a multisystem lysosomal storage disease characterized by progressive central nervous system degeneration manifest as severe intellectual disability (ID), developmental regression, and other neurologic manifestations including autism spectrum disorder (ASD), behavioral problems, and sleep disturbances. Intrathecal ERT and gene therapy are currently under investigation as future therapies. This review provides a historical account of MPS II treatment as well as treatment development with insights into benefits and/or limitations of each specific treatment.Įxpert opinion: Conventional ERT and HSCT coupled with surgical intervention and palliative therapy are currently the treatment options available to MPS II patients. Treatment approaches differ based on age, clinical severity, prognosis, availability and feasibility of therapy, and health insurance. Clinical trials are being conducted for intrathecal ERT and gene therapy is under pre-clinical investigation. Phenotypes are classified as either attenuated or severe (based on absence or presence of central nervous system impairment, respectively).Īreas covered: Current treatments available are intravenous enzyme replacement therapy (ERT), hematopoietic stem cell transplantation (HSCT), anti-inflammatory treatment, and palliative care with symptomatic surgeries.
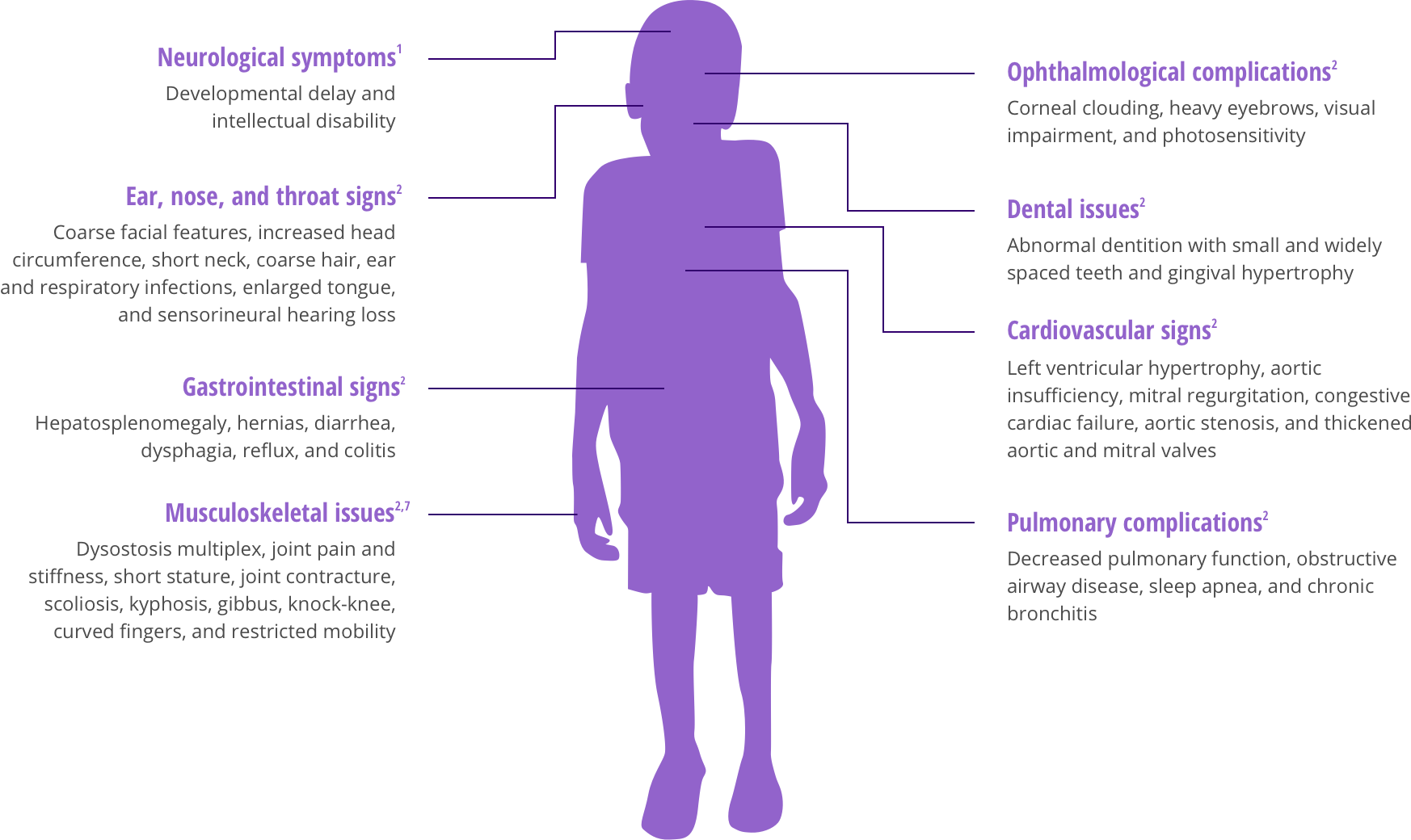
MPS II is both multi-systemic and progressive. IDS deficiency leads to primary accumulation of dermatan sulfate (DS) and heparan sulfate (HS).
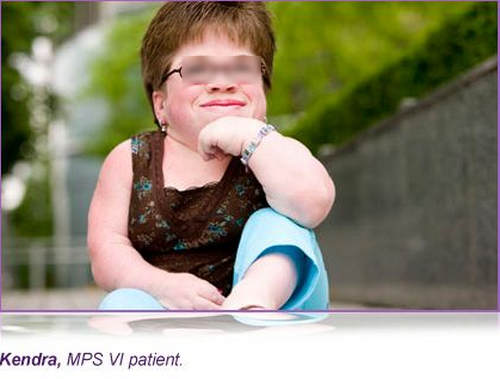
Introduction: Mucopolysaccharidosis Type II (MPS II Hunter syndrome) is an X- linked lysosomal storage disorder caused by a deficiency of iduronate-2-sulfatase (IDS).
